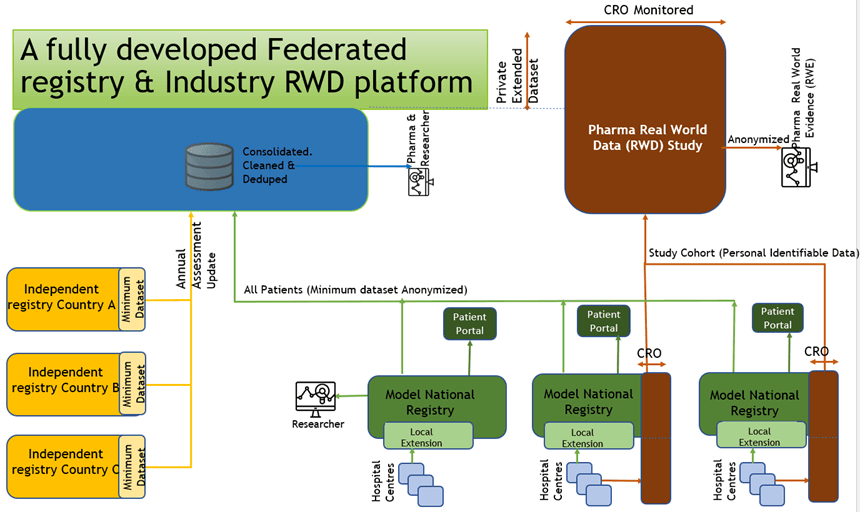
This is the default registry for countries without a sustainable well-established registry. These would typically be smaller countries (for example within Scandinavia or Benelux). The registry is based on:
• Static data collected when the patient enrols in the registry.
• Encounter data which measures the patient’s treatment and outcomes over time.
Encounters can be either:
• A single annual assessment.
• Multiple date-based assessments recorded at each clinical encounter.
A small number of fields (the Minimum Dataset) are defined by the Registry Scientific Working Group as mandatory. A record cannot be completed until data is entered for all mandatory fields.
It is important to keep in mind that registries are completed by “busy doctors under pressure”. Care must be taken to reduce the mandatory data fields in the annual assessment to a minimum.
The Model Registry should never be designed as an electronic version of a paper form. Many paper-based annual assessment reports are completed in the last days of the year with no opportunity to check missing and inconsistent data.
In contrast to a passive paper-based data collection, an online registry is active and can assist doctors during a patient visit. Data validations and prior entry prompts (for example prior medication and dosage) will reduce frequency of data errors.
An online registry will support a much more rigorously defined protocol where the clinician could be alerted if for example there was an unusual change in blood pressure from a prior encounter.
The Model registry can support separate patient pathways to reflect different patient needs. A seriously ill patient needing multiple hospital encounters over a year will generate a lot more relevant data that a healthy patient – however data from both cohorts will need to be fully interoperable. Specifically, a process has to be defined to summarise multiple encounters into one annual summary.